Vaccine-Induced Immune Thrombotic Thrombocytopenia (VITT) Following Adenovirus Vector COVID-19 Vaccination: Interim Guidance for Healthcare Professionals in Emergency Department and Inpatient Settings
Authors:Menaka Pai, Nathan M. Stall, Michael Schull, Katherine J. Miller, Fahad Razak, Benjamin Chan, Allan Grill, Noah Ivers, Antonina Maltsev, Ayodele Odutayo, Brian Schwartz, Michelle Sholzberg, Robert Steiner, Sarah Wilson, Ullanda Niel, Peter Jüni, Andrew M. Morris on behalf of the Drugs & Biologics Clinical Practice Guidelines Working Group and the Ontario COVID-19 Science Advisory Table
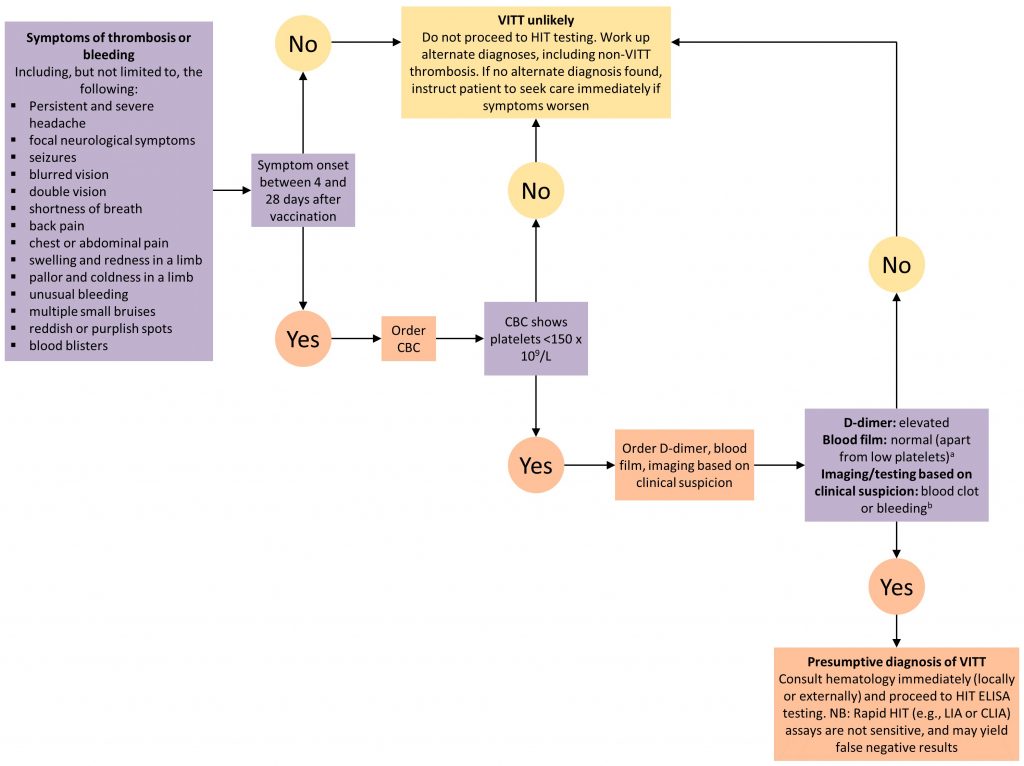
Figure 1. Decision Tree for Diagnosing and Ruling Out VITT
aBlood film to rule out platelet clumping as a cause of low platelet count; bNot all cases of VITT initially present with a clot or bleeding. Patients with all of the features of presumptive VITT (low platelets, high D-dimer, presenting 4 to 28 days post-vaccination) but NO blood clot or bleeding merit hematology consultation (locally or externally) to consider starting treatment until the results of confirmatory testing are available. VITT, vaccine-induced immune thrombotic thrombocytopenia. CBC, complete blood count. HIT, heparin induced thrombocytopenia. ELISA, enzyme linked immunosorbent assay. LIA, latex immunoturbidometric assay. CLIA, chemiluminescent immunoassay.
What do we know so far?
Adenoviral vector COVID-19 vaccines, including the AstraZeneca/COVISHIELD vaccine and the Janssen/Johnson & Johnson vaccine, are associated with immune thrombosis that is similar to heparin-induced thrombocytopenia (HIT). Women and young people appear to be slightly overrepresented in reported cases, and thrombosis seems to occur 4 to 28 days after vaccination. Affected individuals have antibodies targeted against platelet factor 4 (PF4) that induce massive platelet activation, reducing the platelet count and causing thrombosis.1–3 This phenomenon is similar to HIT, but, unlike HIT, VITT does not require heparin as a trigger. It has been referred to as Vaccine-Induced Immune Thrombotic Thrombocytopenia (VITT), Vaccine-Induced Prothrombotic Immune Thrombocytopenia (VIPIT), and Thrombosis Thrombocytopenia syndrome (TTS). In this Science Brief, the term VITT will be used. Published estimates of the incidence of VITT range from 1 case per 26,000 to 1 case per 127,000 doses of AstraZeneca/COVISHIELD administered.3–6 There have been few reported cases of VITT with the Janssen/Johnson & Johnson vaccine thus far, so it is challenging to calculate a precise frequency, but the incidence of VITT appears to be approximately 1 case per 500,000 vaccine doses administered.7
What symptoms should make you suspect VITT?
Patients with VITT may present with cerebral sinus vein thrombosis (CSVT), or with other arterial or venous clots or bleeding. Some symptoms make it more likely that a patient has VITT: persistent and severe headache, seizures, or focal neurological symptoms including blurred or double vision (suggesting CSVT or arterial stroke); shortness of breath; severe chest, back, or abdominal pain (suggesting pulmonary embolism, acute coronary syndrome, abdominal vein thrombosis, or adrenal hemorrhage); unusual bleeding, bruising, petechiae, or blood blisters (suggesting thrombocytopenia or disseminated intravascular coagulation); or limb swelling, redness, pallor, or coldness (suggesting deep vein thrombosis or acute limb ischemia). VITT seems to occur between 4 to 28 days post-vaccination. Symptoms that begin in this time frame should raise the clinical suspicion of VITT.
How do I diagnose VITT? How do I rule it out?
Clinicians should ask patients about their COVID-19 vaccine history and should draw a complete blood count (CBC). VITT is unlikely if symptom onset falls outside of the 4 to 28 day time frame OR if the platelet count is ≥ 150 x 109/L.8–12 VITT is more likely if symptom onset falls within the 4 to 28 day time frame AND the platelet count is < 150 x 109/L.
Patients with suspected VITT should go on to have a D-dimer level and a blood film drawn. They should also have diagnostic imaging to investigate for arterial and/or venous blood clots or bleeding, based on their clinical symptoms. Unenhanced CT brain is a reasonable first diagnostic imaging test if CSVT is suspected, given CSVT’s nonspecific clinical presentation and the importance of ruling out alternate diagnoses. However, CSVT can only be ruled out with both parenchymal imaging and vascular imaging, with a CT head / MR head and a CT venogram / MR venogram.13 When clinical suspicion of CSVT is high, this potential diagnosis should be investigated urgently with same-day neuroimaging. In centres where imaging is not emergently available and there is a high clinical suspicion of VITT, appropriate treatment should be initiated while imaging is arranged.
Thrombocytopenia with an elevated D-dimer, a normal blood film (apart from thrombocytopenia), and confirmation of a blood clot or bleeding on diagnostic imaging/testing makes the diagnosis of VITT presumptive. VITT can also manifest with a low fibrinogen as evidence of more disseminated coagulation consumption.
The confirmatory diagnosis of VITT is made by using tests that are also used for HIT, but with some important caveats. This testing should be done even if the patient has had no previous exposure to heparin. Like HIT, VITT testing involves two steps: identification of antibodies against the complex of platelet factor 4 and heparin; and confirmatory functional testing of the antibodies’ ability to activate platelets.14,15 The HIT ELISA (enzyme linked immunosorbent assay) antibody test appears to be sensitive for VITT because of specific parameters of the assay that also make it applicable to VITT; if it is positive, VITT is confirmed, and if it is negative, VITT is unlikely.14
Importantly, other HIT antibody tests, like the Rapid HIT tests (e.g., latex immunoturbidometric assay (LIA), chemiluminescent immunoassay (CLIA) are not sensitive for VITT, and are likely to yield false negative results. It is important to consult with a hematologist, and the testing laboratory, to ensure that an ELISA method is used for antibody testing. A number of large laboratories test for HIT antibodies using the ELISA method in Ontario, but only one laboratory in Canada performs confirmatory functional testing (the McMaster University Platelet Immunology Laboratory). Therefore, presumptive VITT should prompt an urgent hematology consultation (in person, virtually, or by phone) to arrange HIT ELISA testing, initiate confirmatory functional testing at the national reference laboratory, and start appropriate empiric treatment of blood clots (see below). All testing (HIT ELISA and functional testing) must be sent prior to administration of IVIG treatment, as IVIG can interfere with the results.14
How do I treat VITT?
Patients with presumptive and confirmed VITT should be treated similarly to HIT. Treatment should ideally occur in an emergency department or hospital setting, where the patient can be closely monitored.

Summary Box. Treating Patients with Presumptive or Confirmed VITT
*Platelet transfusions could theoretically worsen the clotting; if patients present with a life-threatening bleed, platelets should only be transfused under the guidance of a hematologist.
Until VITT has been ruled out, anticoagulation with heparin (unfractionated heparin and low molecular weight heparins) should be avoided. Platelet transfusions should not be given.
Alternative anticoagulants that are safe to use in HIT, and likely safe to use in VITT, include direct thrombin inhibitors and factor Xa inhibitors. Direct oral factor Xa inhibitors (e.g., rivaroxaban, apixaban, edoxaban) should be started empirically while awaiting further advice from a hematologist; these agents are used in the treatment of HIT. The dose of direct oral factor Xa inhibitor is identical to the dose used to treat uncomplicated deep vein thromboses. If the patient has severe renal impairment that makes direct oral anticoagulants unsafe, or is too unwell or unstable to take oral medication, advice from a hematologist should be sought to guide use of parenteral anticoagulants that are safe to use in HIT (e.g., argatroban). In pregnant or lactating patients with presumptive or confirmed VITT, direct oral factor Xa inhibitors are not recommended; advice from a hematologist should be sought to guide use of safe non-heparin anticoagulants.
In patients with presumptive or confirmed VITT, it is important to dampen the prothrombotic response and increase platelet counts with intravenous immunoglobulin (IVIG). Administration of high dose IVIG (1 g/kg of actual body weight daily for at least two days) is appropriate and can be guided by the consulting hematologist.
Patients with all of the features of presumptive VITT (low platelets, high D-dimer, symptoms starting 4 to 28 days post-vaccination) but who have NO evidence of thrombosis may have an early VITT syndrome. In these patients, it is important to consult hematology and consider starting anticoagulation and/or IVIG until the results of confirmatory laboratory testing are available.
Is VITT a reportable event?
All suspected adverse events following immunization (AEFI), including thrombosis, thrombocytopenia, and both presumptive and confirmed VITT, should be reported using the provincial AEFI form and sent to the local Public Health Unit. More information on how to report AEFIs can be found on the Public Health Ontario website. Ontario conducts vaccine surveillance safety in collaboration with the Public Health Agency of Canada, and prompt reporting is essential to learn more about this rare but serious thrombotic phenomenon.
